Secure Storage (21 CFR § 1301.75)
Controlled substances must be stored in "securely locked, substantially constructed cabinets."
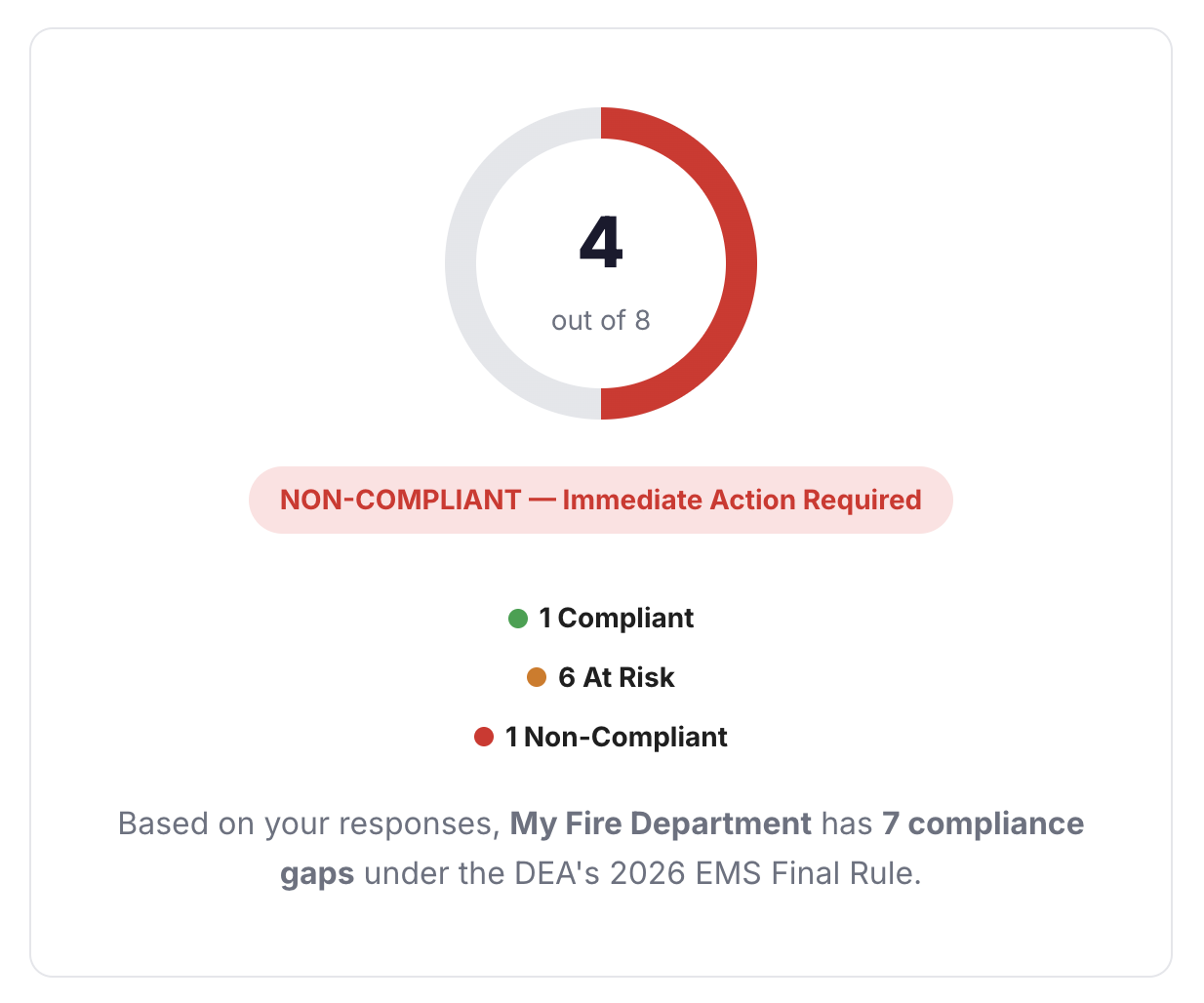
Paper logs and manual tracking consume valuable time, create compliance gaps, and put your medical license at risk.
Hours wasted on manual documentation every week
Mistakes in logs create audit vulnerabilities
Compliance gaps threaten your medical license
Manual tracking delays detection of critical issues
doses of EMS drugs are diverted
EMS vehicles unknowingly carry expired drugs
of EMS staff self-report the abuse of controlled substances
Patient risk. Staff risk. Fines and licensing exposure.
Breaking down the key regulations and what they mean for your organization
Controlled substances must be stored in "securely locked, substantially constructed cabinets."
Maintain complete records of all controlled substance transactions, acquisitions, and disposals.
Immediate notification to DEA within one business day of discovering theft or significant loss.
Limit access to controlled substances to authorized personnel only. Track who, when, and what.
Biennial inventory of all controlled substances. Maintain ongoing inventory records.
Systems and procedures to prevent, detect, and investigate drug diversion.
HIPAA requires unique user identification and emergency access procedures.
Hardware, software, and procedural mechanisms to record and examine access to ePHI.
ePHI must not be improperly altered or destroyed.
Technical security measures to guard against unauthorized access during electronic transmission.
Navigate Narcotics Compliance With Confidence